Donating a little bit of time can make a big impact. On Nov. 21, members of the Department of Computational and Systems Biology (CSB) spent the day volunteering at Cribs for Kids.

Menu

Donating a little bit of time can make a big impact. On Nov. 21, members of the Department of Computational and Systems Biology (CSB) spent the day volunteering at Cribs for Kids.

Emeritus Professor Hagai Meirovitch returned to the University of Pittsburgh for a visit last week.

Have you ever wondered where a master’s degree in Computational Biomedicine and Biotechnology (CoBB) will take you? Learn more about the data science profession.

Alumnus Murat Can Cobanoglu works as a staff bioinformatics scientist at Natera. Using a blood sample, Cobanoglu’s team can tell whether a patient has cancer or not.

The University of Pittsburgh Department of Computational and Systems Biology welcomed leading genomics researchers from across the country to Pitt’s University Club on Oct. 7–8 for the 2025 Centers of Excellence in Genomic Science (CEGS) Annual Meeting.

Swapnil Keshari was looking for a way to de-stress from his PhD studies when he stumbled upon YouTube reels about aviation. After watching those first few videos about the freedom of flight, he knew he wanted to earn his private pilot license.

The Gur Lab has unveiled a fast, practical way to read molecular “movies” and pinpoint the forces that make proteins stick together or push apart.

Are you a health care student eager to amplify your impact? Imagine a career where clinical expertise merges with data-driven innovation—where you’re not just practicing medicine but shaping its future.
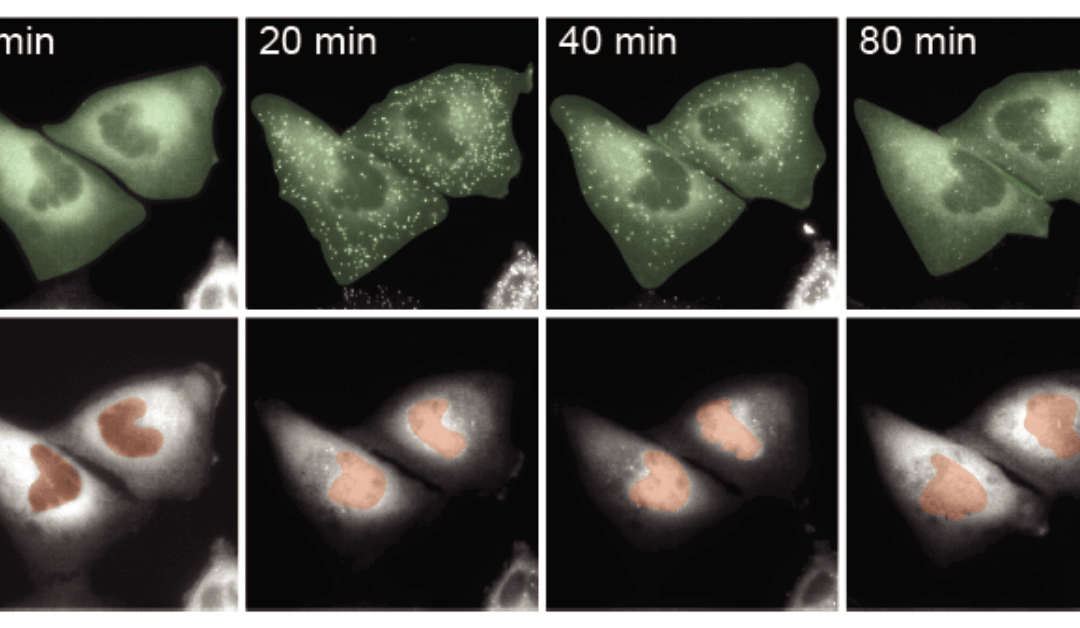
In the complex world inside our bodies, timing can be everything. A new study from Steven Smeal and Robin E.C. Lee reveals the timing of molecular signals can change how cells respond to their environment, with potential implications for cancer treatment and drug discovery.

When Chakra Chennubhotla faced questions in cancer research that existing technologies couldn’t answer, he decided to help build new solutions. That decision ultimately led him to co-found PredxBio, a company at the forefront of AI-driven spatial biology.