Title: Role of a Novel Mitotic 4E-BP1 Protein Isoform in Cellular Transformation
PI: Yuan Chang
Co-I: Robin Lee and Patrick Moore
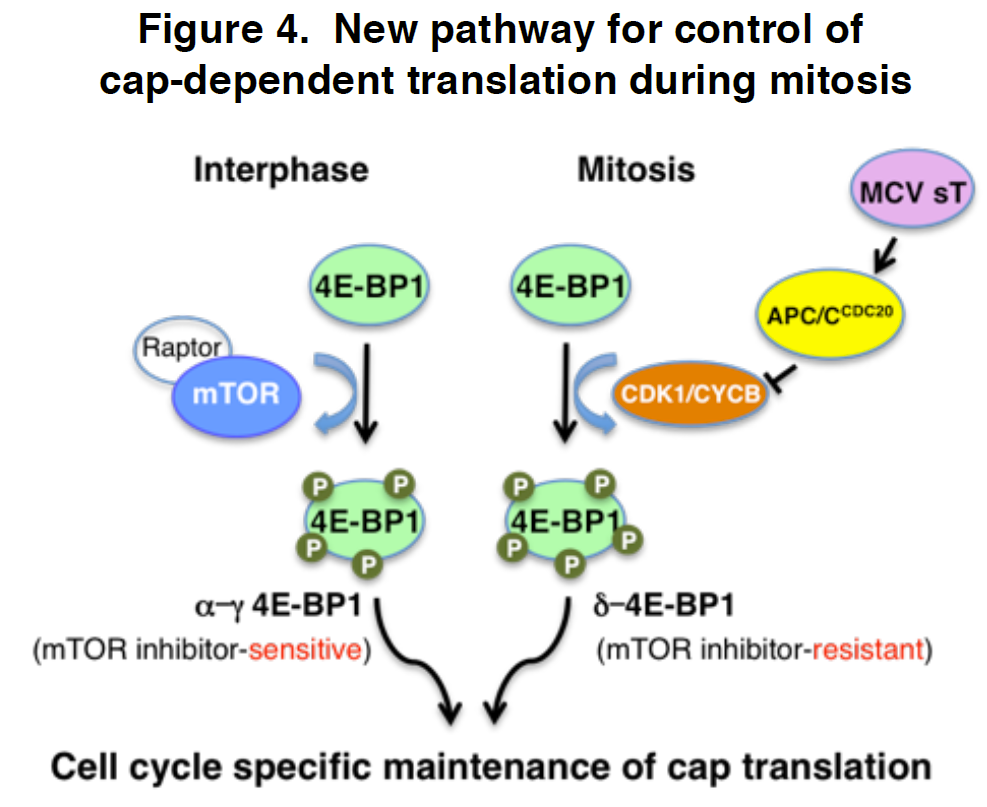
4E-BP1 is the primary gatekeeper for cancer cell cap dependent protein translation. It is directly targeted by mTOR kinase during interphase. We have found that CDK1/CYCB1 substitutes for mTOR during mitosis to phosphorylate 4E-BP1 generating a novel phosphorylation mark at serine (S) 83 that is not present when mTOR phosphorylates 4E-BP1. Unlike other 4E-BP1 phospho-isoforms, phospho-4E-BP1-S83 preferentially localizes to mitotic centrosomes as well as being diffusely distributed in a speckled pattern in the nucleo/cyto-plasm. A mutant form of 4E-BP1 that is unable to be phosphorylated at S83 partially reverses cell transformation caused by the Merkel cell polyomavirus (MCV) small T oncoprotein. This is particularly interesting since discovery of a novel pathway targeted by a tumor virus has always led to discovery of the same pathway being altered in non-infectious cancers. We developed a new phosphospecific antibody to p4E-BP1S83 that allows us to uniquely identify mitosis-related 4E-BP1 phosphorylation and determine its function in cancer cells. With this antibody, we will first survey phosphor-4E-BP-S83 expression in TCGA cancer tissues and anticipate that this approach will be a sensitive measure for activated CDK1 circuits and will provide unique data on cancer-type specific severity. We will next examine the biology of p4E-BP1S83 by identifying S83 phosphospecific effects on mitogenesis and by examining 4E-BP1’s potential role in regulating translation of specific transcripts during mitosis using ribosomal profiling and novel quantitative single cell imaging techniques. Finally, we will generate knock-in mutant mouse models of inactivated and phosphomimetic mitotic 4E-BP1 to determine its role in tumor susceptibility. Our specific aims will advance our fundamental understanding of how a mitosis-specific, hyperphosphorylated form of 4E-BP1 functions in normally cycling cells and how its dysregulation in cancer cells may contribute to human malignancies.
 |
 |
 |
| PI: Yuan Chang, MD | Co-I: Robin Lee, PhD | Co-I: Patrick Moore, MD, MPH |
